Project Title: Development of Capsule Type Respiratory Products
Client Name: Teva Pharmaceuticals
Unveiling the Future of Dry Powder Inhalers through Advanced Simulation and High-Speed X-Ray Imaging
Introduction:
Teva, a global leader in generic and speciality medicines, has made groundbreaking strides in the development of its new line of capsule dry powder inhalers. Leveraging state-of-the-art technologies like Computational Fluid Dynamics (CFD) and High-Speed X-ray imaging, this project offers unparalleled insights into inhaler performance, laying the groundwork for superior inhaler designs that promise improved patient outcomes.
The Challenge:
Previous research for Teva devices faced a daunting roadblock: the inability to validate CFD models of particle flows with real-time imaging. Traditional optical techniques fell short in capturing the intricate internal dynamics of the inhaler, highlighting the need for innovative solutions.
Our Approach: A Comprehensive Investigation
The research project was structured into five critical tasks:
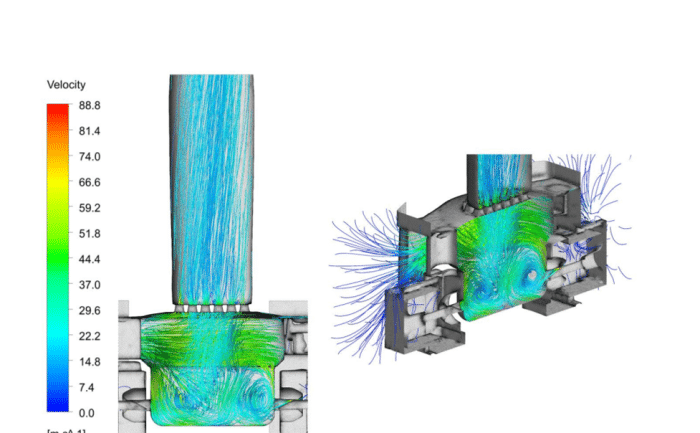
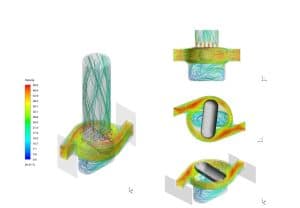
XMT and CFD Inhaler Modelling: We developed a reliable CT CFD model of the inhaler, utilising software tools such as VGStudio Max, MeshLab, and SpaceClaim.
CFD Modelling of Capsule Movement: Advanced simulations were conducted to explore the capsule’s influence on pressure drop distribution and angular velocity.
Analysis of Capsule Dose Evacuation: Particle motion methodologies were incorporated to accurately predict capsule evacuation.
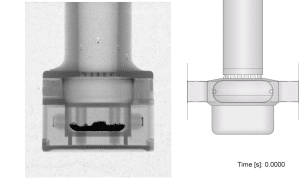
High-Speed X-Ray Imaging: Employing custom-developed imaging techniques, we achieved real-time visualisation of the inhaler’s internal mechanisms.
Custom Particle Model: Utilising ANSYS Fluent UDF for particle force history monitoring, we gained insights into particle dynamics, including wall collisions and turbulence effects.
Results and Validation:
High-speed X-ray imaging allowed us to validate our CFD models effectively. The strong correlation between simulation and real-world observation underscored the model’s accuracy in predicting realistic capsule and particle motion. This successful validation marks a significant milestone in the understanding of the clinical performance of dry powder inhalers.
Impact and Future Prospects:
The methodologies and findings from this project not only deepen our understanding of inhaler dynamics but also serve as a stepping stone for future research. By achieving these innovative results, Teva stands poised to lead the industry in the development of more effective inhaler designs, thus improving patient outcomes.
Mr Dan Buck Senior Director at TEVA said “Teva have had a close working relationship with SEAM for over 15 years and this project is another example of what can be achieved with a collaborative mind-set”
Dr Ramesh Raghavendra Centre Director at SEAM said “We take pride in collaborating with Irish based industries and this innovation partnership project with Teva is a great example of what happens when you establish strong relationship and get talented people across the aisles meet together with marketing leading technology that is on offer ”
If you would like to learn more about the innovation partnership Programme please click here or contact us at SEAM to discuss the benefits of an innovation partnership.
Download PDF Version Here innovation partnership TEVA & SEAM (4)